Understanding Regulatory Affairs in Biotechnology
An India-Focused Guide
Regulatory affairs in biotechnology ensures safe, effective products—like vaccines, gene therapies, and diagnostics—reach patients. In India, the Central Drugs Standard Control Organisation (CDSCO) leads under the Drugs and Cosmetics Act, 1940, alongside global bodies like the U.S. FDA and EMA. Picture it as the bridge between lab breakthroughs and real-world use: it blocks unsafe innovations and accelerates promising ones, vital for India’s $100B+ biotech industry.
This guide outlines the process with an Indian lens, highlighting pros, cons, and challenges. It’s key for bioinformatics, biomarkers, and India’s vaccine hub status.
What Is Regulatory Affairs in Biotech?
Regulatory professionals manage compliance: preparing submissions, engaging agencies, and enforcing standards. Biotech biologics—from living cells like monoclonal antibodies or CRISPR tools—demand specialized rules.
In India, CDSCO’s Central Licensing Authority (CLA) oversees via Form CTS/CTE for trials; global ICH guidelines align with DCGI approvals.
Key tasks:
· File Investigational New Drug (IND)-equivalent permissions for trials.
· Uphold Good Practices: GLP (labs), GCP (clinics), GMP (manufacturing).
· Handle post-approval changes, like safety label updates.
Pros:
· Protects public health, as in India’s fast COVID vaccine rollouts.
· Fast-tracks via CDSCO’s priority review cut times to 30 days.
· Boosts exports—India supplies 60% of global vaccines.
Cons:
· Paperwork overload delays startups.
· Evolving rules for gene therapies challenge firms.
· Harmonization gaps with FDA/EMA slow global entry.
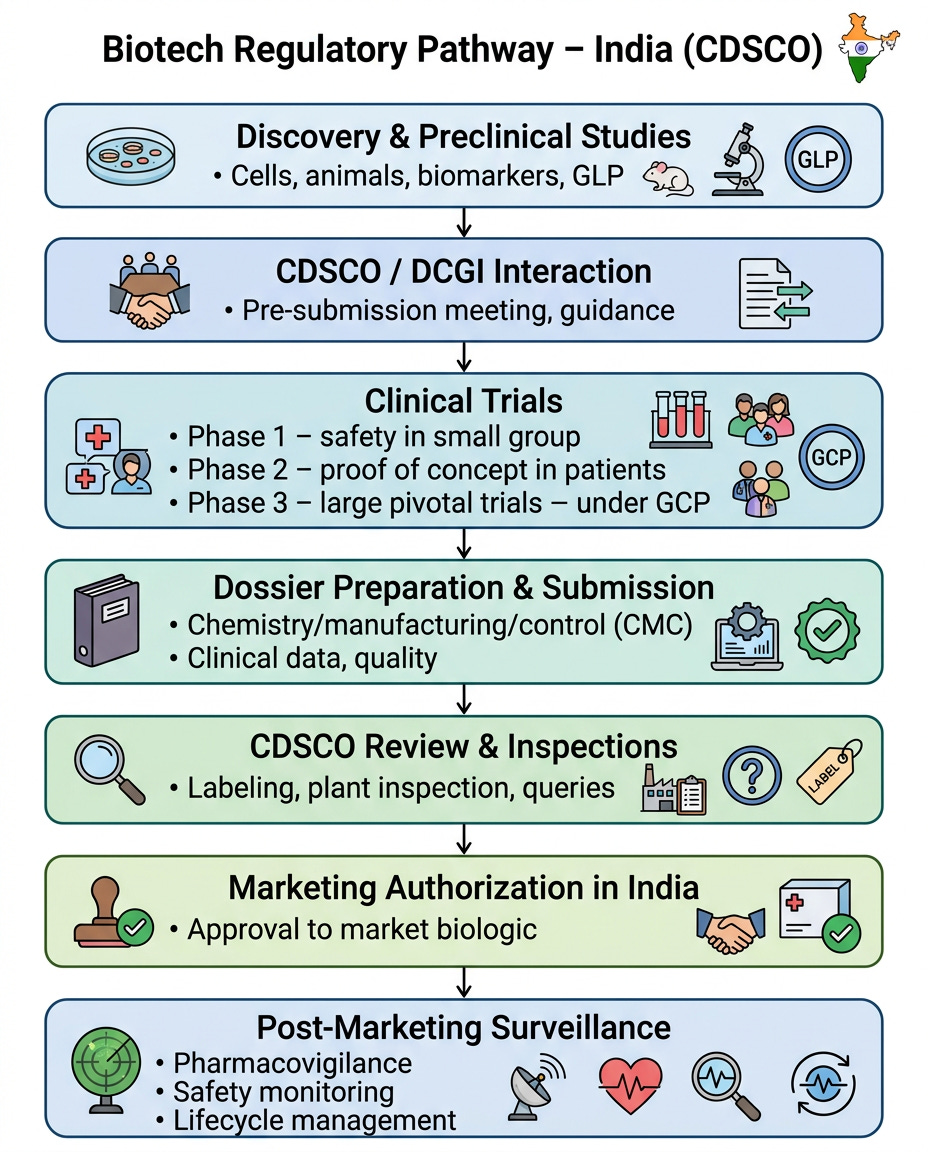
The Drug Approval Pathway: Stage-by-Stage Breakdown
India’s path mirrors global standards but is faster/cheaper: 5-10 years, ₹500-1,500 crore ($60-180M), ~15-25% Phase 1-to-approval success. CDSCO approvals often precede FDA.
1. Preclinical Stage (Discovery to Permission)
Test in cells/animals for safety/efficacy. Goal: CDSCO permission for human trials.
Key activities:
· GLP toxicology/pharmacology.
· Biomarker validation (genomics for early signals).
· GMP scale-up.
Timeline: 2-4 years. Key stats: 70-80% advance; costs ₹50-250 crore; failures from toxicity (40%) or efficacy (30%).
Pros: Low-cost local labs; pre-submission CDSCO meets.
Cons: Animal-human mismatches (80% revisions).
Struggles: Data gaps cause holds (~20%); diverse Indian datasets aid biomarkers.
2. Phase 1: Safety First (Small Human Groups)
Dose 20-100 volunteers for safe ranges/side effects.
Key activities:
· Dose escalation, pharmacokinetics.
· Adverse monitoring.
Timeline: 6-18 months. Success rate: ~70%.
Pros: Quick local recruitment; orphan drug incentives.
Cons: High per-patient costs (~₹10 crore).
Struggles: Immunogenicity (e.g., early biologics); PK variability in diverse populations.
3. Phase 2: Proof of Concept (Targeted Patients)
Efficacy in 100-300 patients; endpoints like biomarker shifts.
Key activities:
· Randomized trials.
· Genomics/proteomics analysis.
Timeline: 1-2 years. Success rate: 30-40%.
Pros: Partnerships (e.g., Bharat Biotech); adaptive designs.
Cons: Rare disease recruitment lags; costs ₹200+ crore (~₹5 crore/patient).
Struggles: Biomarker mismatches (40% attrition; 25% biomarker failures); 6-12 month delays. Example: Indian Alzheimer’s candidates stalled on signals.
4. Phase 3: Pivotal Confirmation (Large-Scale)
Validate in 1,000-3,000 patients, often bridging global data.
Key activities:
· Comparator trials; genotype subgroups.
· Long-term safety.
Timeline: 2-3 years. Success rate: 50-70%.
Pros: Market exclusivity (7-10 years); rolling reviews.
Cons: Costs ₹1,000+ crore.
Struggles: Stats/manufacturing issues (CMC rejections ~15%). Example: Serum’s CAR-T-like Covishield scaled amid global scrutiny.
5. Marketing Approval (Permission to Market)
Submit full dossier; CDSCO reviews in 3-6 months (priority: 30 days).
Key activities:
· Label negotiations; site inspections.
· Risk programs.
Pros: Revenue surge (Covishield: ₹15,000+ crore); biosimilars thrive.
Cons: Post-approval loads.
Struggles: Response letters (10-20%); CMC (15%), labeling (10%). mRNA approvals tested potency.
Post-Approval: Lifecycle Management
Pharmacovigilance via PvPI; changes need CDSCO nods. Pros: Patent extensions. Struggles: Adverse reports (e.g., Covishield clots) trigger updates.
Regulatory affairs powers India’s biotech rise—from Covaxin triumphs to global exports—despite hurdles like costs and data gaps. Careers like Regulatory Affairs Officers (₹10-20L entry) or Managers (₹30L+) suit science grads with RAC/DBT training. Biomarker/FDA experience excels in validation; track CDSCO’s New Drugs Rules and AI updates. Explore CDSCO portal or RAPS India courses
.